Nano Chemistry - Laursen Group

Our research concerns the design and synthesis on fluorescent dyes and molecular materials with new and improved optical properties. Our aim is to understand the relations between molecular structure and optical properties (molecular photophysics), and to use this insight to tailor dyes and materials to be used e.g. in: fluorescent probes and sensors, cell imaging, fluorescence guided surgery, ultra-bright crystals and nanoparticles, organic laser materials and other optical materials applications. In the group we focus on design, synthesis and spectroscopy, and we collaborate with a broad range of physicists, biologists, medical researches, other nanoscience and chemistry groups about the applications and properties of our materials.
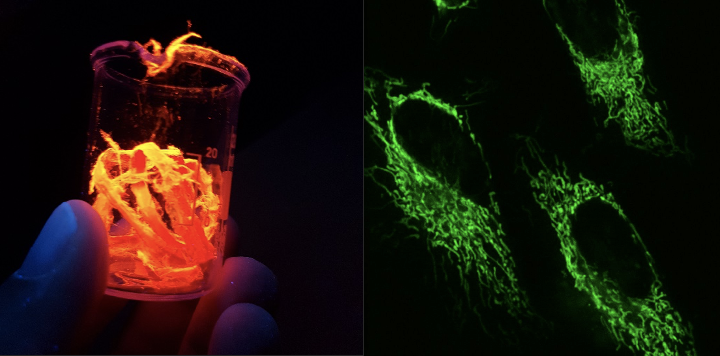
Currently our research is focused in the three main areas outlined below. Furthermore, we take part in many collaborative projects contributing our dyes and materials or our spectroscopic capacity on systems synthesized by collaborators.
Triangulenium dyes
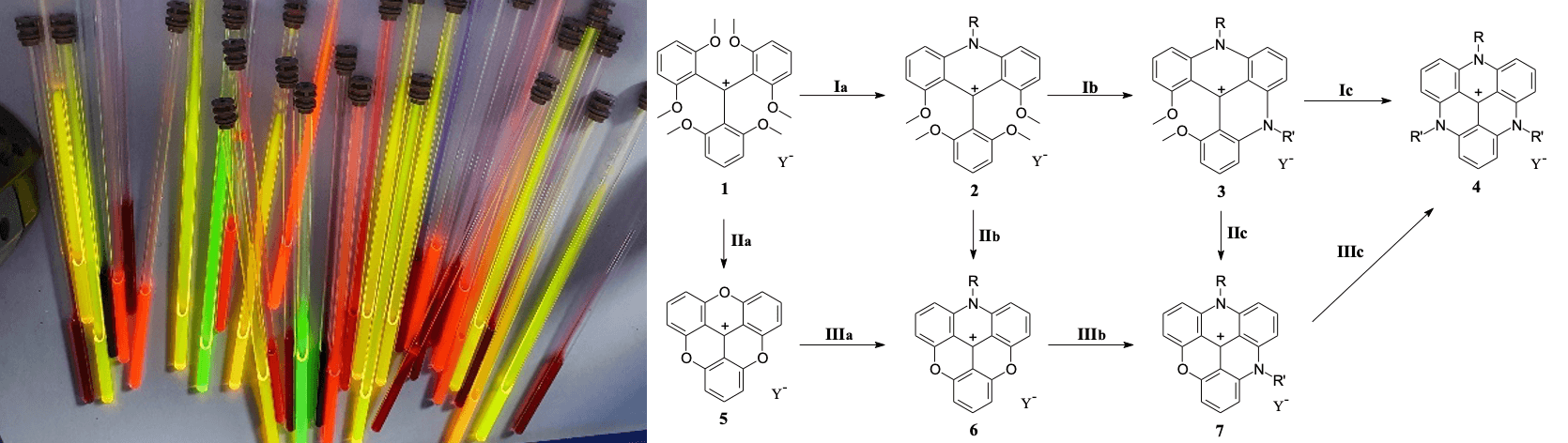
Triangulenium dyes are carbenium ions of exceptional chemical stability, and broad structural diversity owing to the facile synthetic routes developed over the years. We constantly explore new transformations and structural motifs to expand our toolbox of dyes with unique optical properties.
Recent examples of dye development include:
- Utilizing selective chlorination to synthesize new triangulenium”, Journal of Organic Chemistry, 2021, 86, 17002-17010.
- “What is Best Strategy for Water Soluble Fluorescence Dyes? - A Case Study Using Long Fluorescence Lifetime DAOTA Dyes”, Chemistry - A European Journal, 2020, 26, 15969-15976.
- “Synthesis and Properties of Sulfur Functionalized Triarylmethylium, Acridinium and Triangulenium Dyes”, Beilstein Journal of Organic Chemistry, 2019, 15, 2133–2141.
- “Extended triangulenium ions – syntheses and characterization of benzo-bridged dioxa- and diazatriangulenium dyes”, Journal of Organic Chemistry, 2019, 84, 2556-2567.
Photophysics and applications of dyes
The high photo-stability and unique long fluorescence lifetime of the aza/oxa-triangulenium dyes (10-20 ns) provides attractive opportunities in time-resolved imaging and sensing, anisotropy assays and for PET base fluorescent probes/sensors. Some recent examples of such dye applications include:
- “Fluorescent pH Probes based on PET Quenching of Long Fluorescence Lifetime Triangulenium Dyes", ChemPhotoChem, 2019, 3, 233–242.
- “Design, Synthesis, and Time Gated Cell Imaging of Carbon-Bridged Triangulenium Dyes with Long Fluorescence Lifetime and Red Emission”, Chemical Science, 2018, 9, 3122-3130.
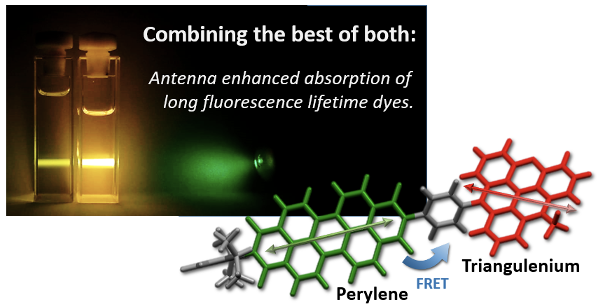
- “Rational design of bright long fluorescence lifetime dyad fluorophores for single molecule imaging and detection”, Am. Chem. Soc., 2021, 143, 1377-1385.
Fluorescent molecular materials and nanostructures:
When organic dyes pack close together in solid state materials or aggregates/nanostructures their optical properties are most often dramatically changed due to electronic coupling between the transition moments, which both gives reis to new optical transition and efficient energy migration in the nanostructures. We are interested in understanding these processes and to develop methods that allow us to organize the dyes in specific structures and thus control the properties.
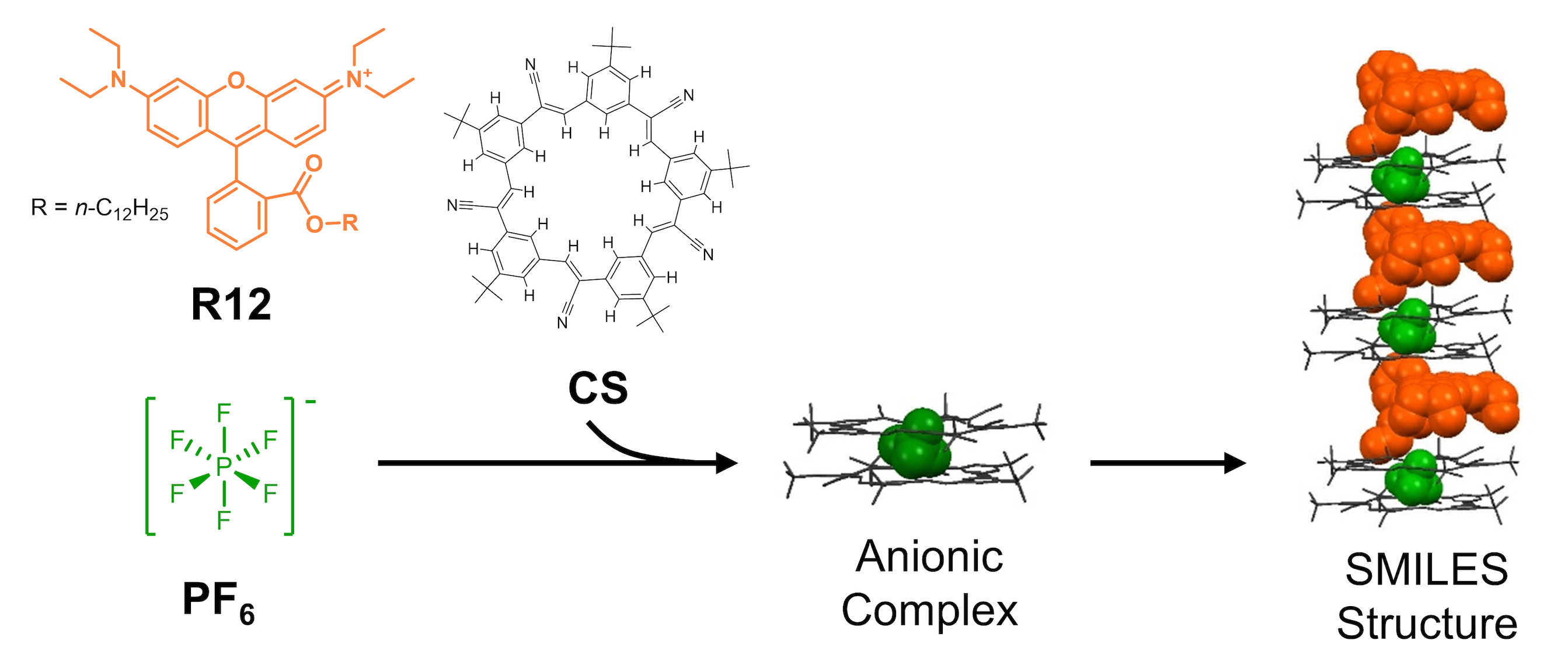
Recently we have in particular worked with an efficient supramolecular self-assembly approach to organize cationic dyes in solid state materials by use of the anion binding macrocycle cyanostar. In the resulting “SMILES” materials the dyes are isolated and retain their intrinsic molecular properties and produces ultra-bright crystals, thin-films, polymer blends and nanoparticles. This work is done in close collaboration with the Flood group at Indiana University. Some recent work on SMILES and dye materials includes:
- “A Universal Concept for Bright, Organic Solid State Emitters - Doping of Small Molecule Ionic Isolation Lattices with FRET Acceptors”, Am. Chem. Soc., 2022
- “Ulta-Bright Fluorescent Organic Nanoparticles based on Small-Molecule Ionic Isolation Lattices”, Angewandte Chemie International Edition, 2021, 60, 9450-9458
- Plug-and-Play Optical Materials from Fluorescent Dyes and Macrocycles", Chem, 2020, 6, 1978-1997.


Email: bwl@chem.ku.dk
Office: CS16 HCØ
Phone: +45 40 43 38 81
Address: Nano-Science Center and Department of Chemistry, Universitetsparken 5, 2100 Copenhagen Ø
Master Students
Andreas Gommesen Nilsson
Amalie Frandsen
Dalma Edit Nánási
Julian Lava
PhD Students
Stine Grønfeldt Stenspil
Jesper Dahl Jensen
Yang Wang
Marko Heine Nowack
Rasmus K. Jakobsen
Postdocs.
Abbey Philip
Kapil Kumar
Contact
Bo W. Laursen
Professor
Office: CS16
Phone: +45 3532 1881
Cell: +45 4043 3881
E-mail: bwl@chem.ku.dk
