Nanoparticles in Electrocatalysis – Pittkowski Group
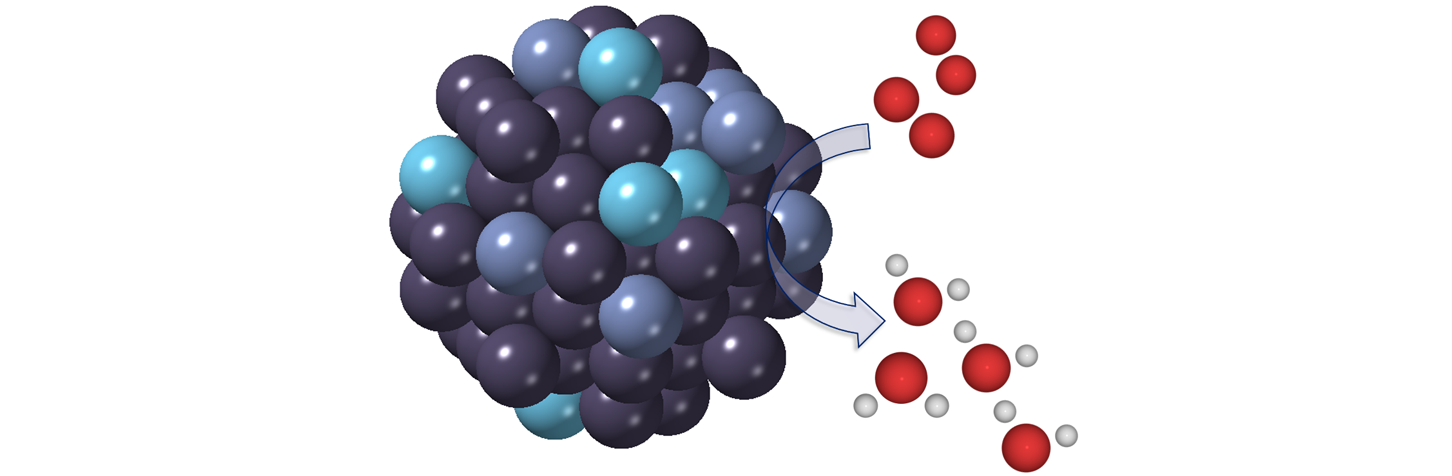
The main research interest of the group is to investigate the relations between nanoparticle structure and catalytic properties. We aim to use this knowledge in developing new nanomaterial catalysts for energy conversion reactions. Our research combines electrocatalysis with nanoparticle synthesis, and advanced characterization techniques. We synthesize nanoparticles of metal alloys and oxides and examine their catalytic behavior within reaction environments, to uncover how the material's structure influences its activity and stability as a catalyst. Understanding the structure and its transformations under reaction conditions is particularly important to develop new, stable catalysts. To study the active catalyst structure we employ synchrotron X-ray techniques, such as X-ray absorption spectroscopy.
Our primary focus lies in electrocatalysis, a key component in conversion and storage technologies for renewable energy. We explore two particular examples: producing emission-free hydrogen through water electrolysis and directly converting chemical energy into electricity in fuel cells.
The group is led by Rebecca Pittkowski, tenure-track assistant professor at the Department of Chemistry, University of Copenhagen.
Electrocatalysis of nanoparticles
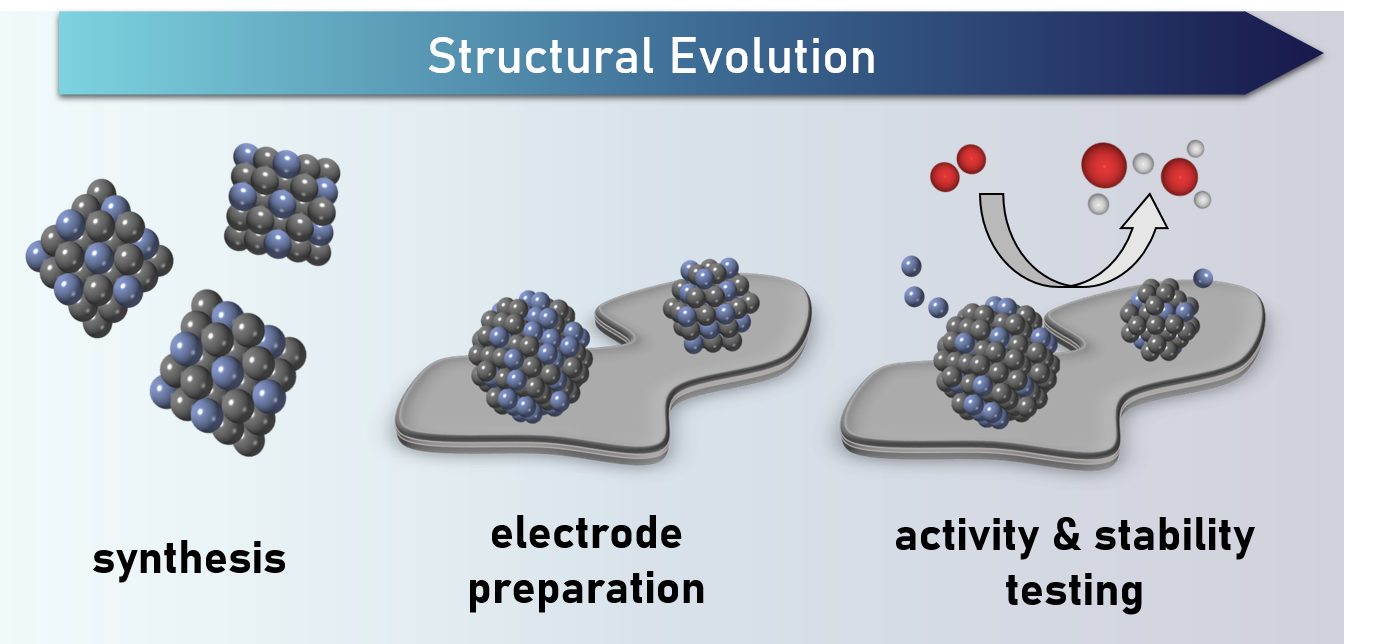
Electrocatalysis is an important factor in moving towards a more sustainable society. Using renewable electricity, we can catalyze redox reactions to transform chemicals and fuels. Our research focuses on the catalysis of key reactions in fuel cells and electrolyzers for energy applications. To achieve a high utilization of the catalyst material, we focus on nanoparticles. These nanostructures possess a very high percentage of surface atoms, which can be active in the catalysis. We synthesize nanoparticles and study their electrocatalytic properties in reduction and oxidation reactions. A key concern in developing new catalysts for us is that the nanoparticles retain their stability under reaction conditions.
X-ray absorption spectroscopy
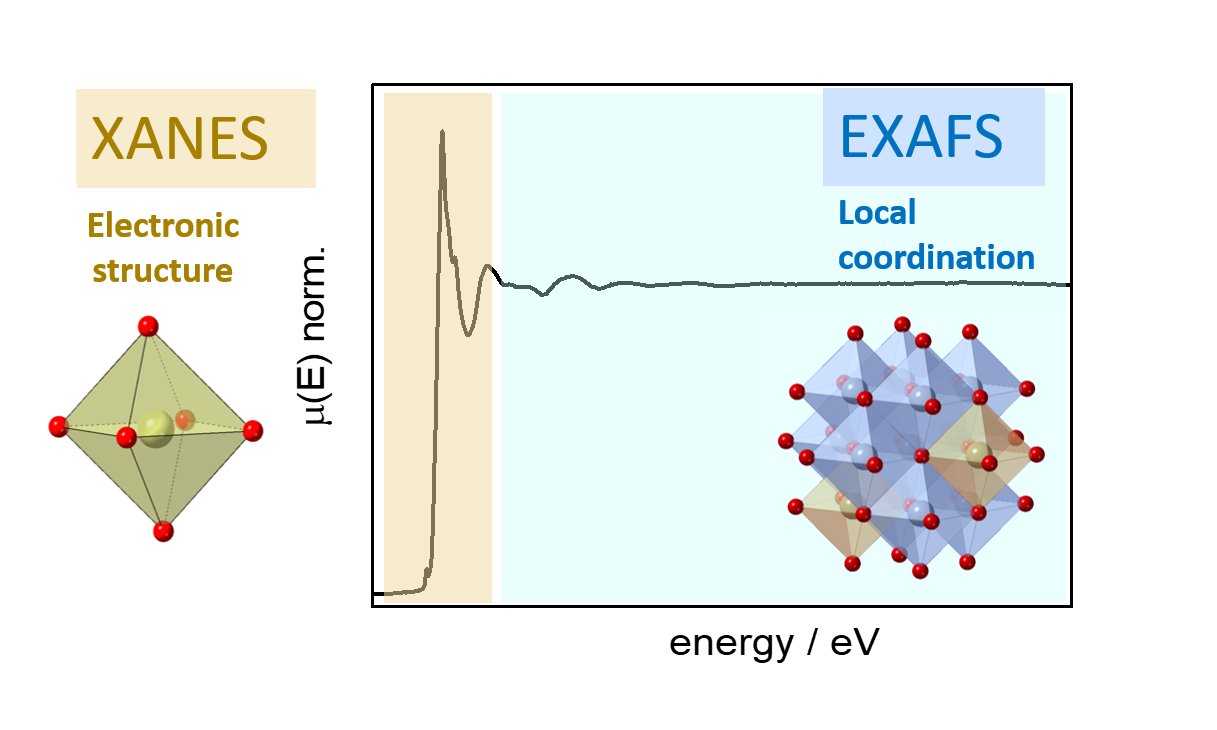
Advanced new characterization methods allow us to obtain complex structural information. We use X-ray absorption spectroscopy (XAS) as a tool to capture the electronic structures and local environments of catalysts. XAS allows us to obtain structural information on nanostructured and disordered catalyst materials. To measure XAS, we use synchrotron X-ray radiation. We can obtain detailed information on the valence states and electronic structures of specific elements with X-ray absorption near-edge structure (XANES), while extended X-ray absorption fine structure (EXAFS) provides information on the local coordination geometry and bonding distances. XAS is a powerful technique for element-specific studies of local geometries. This information is important for us to understand the structure of the active site in catalysis.
Structure of the operating catalysts
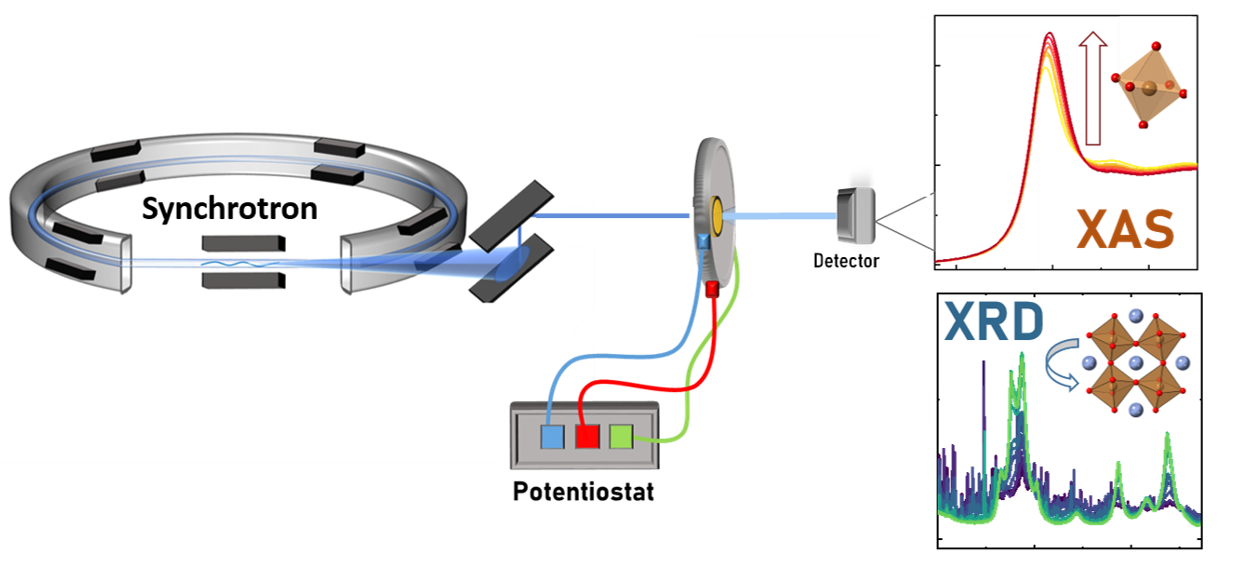
Understanding how structure and properties of materials are related is essential for catalyst development. To achieve this, we need to know the structure of the catalytically active material. This exists only when the catalyst is active. With the high energy X-rays that are available at synchrotron facilities, we can “look into” our electrocatalysis set-up and study the structure of the active catalysts. With in situ and operando XAS techniques, we can establish correlations between electrochemical performance and structural properties. We combine these XAS studies with complementary X-ray based techniques, such as powder X-ray diffraction, to get a complete picture of the catalyst structure during operation. These studies of active catalysts in the reaction environments allows us to also follow the processes that lead to losses in catalyst performance.
Contact
Rebecca K. Pittkowski
Assistant Professor
Office: C106A
Mail: rebecca.pittkowski@chem.ku.dk
Phone: +45 3533 3633
